Surfactants are the main ingredients in every cleanser, shampoo, and face wash.
Whether you’re formulating a gentle sulfate-free shampoo or a foaming facial cleanser, understanding how surfactants work is essential. From removing dirt and excess oil to delivering that satisfying lather, these ingredients can make or break your product’s performance and your skin and hair health.
In this guide, we’ll break down the main types of surfactants, clear up common myths about SLS, and help you choose milder alternatives that align with your formulation goals.
Table of Contents
Surfactants in Skincare and Haircare: What They Are and How They Work
Surfactants (short for surface-active agents) are molecules that allow oil and water to mix. They’re what make your cleanser foam and lift away impurities. A surfactant has a water-loving head and an oil-loving tail. This clever structure grabs onto grime, traps it inside tiny bubbles called micelles, and rinses it all away.
While some surfactants (like SLS) have earned a reputation for being harsh, many modern options are designed to be mild, biodegradable, and suitable even for sensitive skin and scalp. Understanding how these ingredients function is the first step to formulating products that clean effectively without causing irritation or dryness.
What Are Surfactants?
Surfactants are the backbone of almost every cleansing product, from facial washes to solid shampoo bars. But what exactly makes them so effective?
At a molecular level, surfactants have a split personality:
- One end (the hydrophilic head) loves water.
- The other end (the lipophilic tail) is drawn to oils and dirt.
When you apply a cleanser, the tails latch onto sebum, makeup, and debris, while the heads stay attracted to water. As you rinse, these surfactant molecules bundle up the grime into tiny spheres (called micelles) and lift it all away effortlessly.
This process is why a surfactant-rich formula can leave skin feeling fresh and hair feeling clean. But the type and concentration of surfactant you choose makes all the difference between a gentle, balanced product and one that strips away too much of your natural oils.
How Surfactants Cleanse: The Micelle Mechanism
Imagine surfactants as tiny magnets with two poles. The oil-loving tails surround dirt and oils on your skin and hair. Together, they form micelles, miniature cleaning squads, that suspend impurities so they can be rinsed off with water.
This micelle action is why surfactants are so effective in both skincare and haircare. They don’t just spread water over your skin; they actually dislodge and encapsulate unwanted debris. That’s also why even a small amount of surfactant can create a powerful cleansing effect.

Above diagram shows how surfactants organize themselves around oils and debris. The hydrophobic tails face inward, trapping impurities, while the hydrophilic heads point outward toward the water.
This micelle formation is what allows cleansers and shampoos to effectively remove dirt without the need for harsh scrubbing.
Why Surfactants Matter in Skincare and Haircare Formulations
Choosing the right surfactant isn’t just about foam or marketing claims. The selection directly affects:
- Cleansing strength: How thoroughly your product removes oils and buildup.
- Mildness: Whether it disrupts the skin barrier or scalp microbiome.
- Foam texture: From rich and creamy to light and bubbly.
- Rinseability: How easily it leaves the hair or skin clean without residue.
Formulators need to strike a balance between effective cleansing and maintaining the health of the skin and hair. Especially in sensitive skin products or color-treated hair care, this balance is critical.
The Different Types of Surfactants
Not all surfactants are created equal. Each type has its own personality; some are strong cleansers, while others are mild and conditioning. Let’s break them down so you can see which options best fit your formulation goals.
Surfactants are broadly categorized by the charge they carry when dissolved in water. This charge influences how they interact with oils, dirt, and the skin or hair surface.
Understanding these main types: anionic, nonionic, amphoteric, and cationic, helps you choose the right combination for effective yet gentle cleansing.
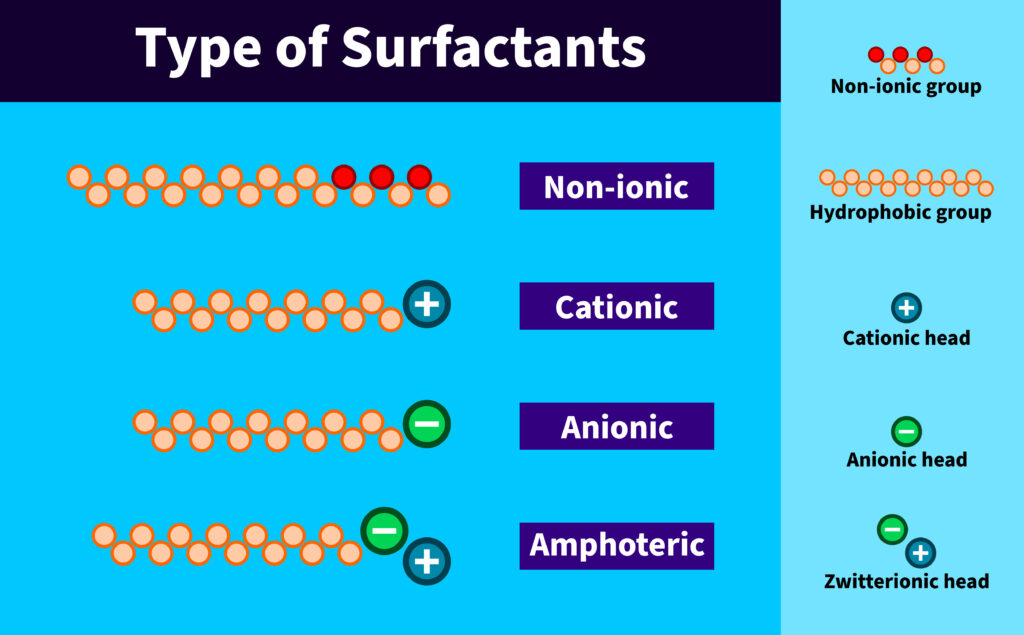
Each surfactant type has distinct characteristics. Anionics offer strong cleansing, nonionics provide mildness, amphoterics balance foam and gentleness, and cationics deliver conditioning. Selecting the right blend is essential for creating well-rounded skincare and haircare formulations.
Anionic Surfactants: Strong Cleansing Power
Anionic surfactants carry a negative charge when dissolved in water. This makes them highly effective at removing oils, dirt, and styling residues. They’re often the primary cleansers in shampoos and body washes.
Examples you’ll often see:
- Sodium Lauryl Sulfate (SLS): Famous, and sometimes infamous, for its powerful cleansing and foaming. We’ll cover SLS myths and facts in detail below.
- Sodium Coco-Sulfate (SCS): Derived from coconut oil; a bit milder than SLS but still delivers big, bubbly lather.
- Sodium Cocoyl Isethionate (SCI): A sulfate-free option known for its creamy foam and gentle action. Popular in solid shampoos.
Quick Note on SLS:
Although SLS is one of the most debated surfactants, it’s not inherently dangerous. Still, many formulators prefer alternatives to minimize irritation. (For a deeper look at SLS and when to avoid it, see “The Truth About SLS” below.)
💡 Related Reading:
Want to dive deeper into why Sodium Cocoyl Isethionate (SCI) is such a game-changer for solid shampoo bars? Learn about its chemistry, formulation tips, and common mistakes to avoid in our dedicated guide.👉 Read: Sodium Cocoyl Isethionate – Why It’s the Hero of Solid Shampoo Bars
Nonionic Surfactants: Mild and Versatile Cleansers
Nonionic surfactants have no electrical charge, making them gentler on skin and hair. They’re often used in baby shampoos and sensitive skin cleansers.
Common examples:
- Coco Glucoside: Plant-based, biodegradable, and known for stabilizing foam while staying mild.
- Decyl Glucoside: Similar to coco glucoside but slightly less viscous; excellent for low-irritation cleansers.
- Lauryl Glucoside: Creamy texture, often used to improve the feel and thickness of formulas.
Nonionic surfactants are also excellent “co-surfactants” to balance out stronger cleansers.
💡 Related Reading:
Curious about how Alkyl Polyglucosides (APGs) like Coco Glucoside and Decyl Glucoside really work in haircare? Learn how to formulate with them effectively, even in solid bars, and discover why formulators love these gentle, biodegradable surfactants.
Amphoteric Surfactants: Balancing Mildness and Foam
Amphoteric surfactants adapt their charge based on pH, making them flexible team players. They boost foam and reduce irritation when combined with anionics.
Key examples:
- Cocamidopropyl Betaine: One of the most popular mild surfactants worldwide. Adds gentle cleansing and helps stabilize lather.
- Disodium Cocoamphodiacetate: Often chosen for ultra-gentle products like baby washes.
When you want balanced cleansing without over-drying, amphoterics are invaluable.
Cationic Surfactants: Conditioning and Detangling
Cationic surfactants carry a positive charge, making them stick to the negatively charged hair shaft. They don’t foam but are brilliant for conditioning and reducing static.
Common cationic agents:
- Behentrimonium Methosulfate (BTMS): Adds slip and softness—ideal for conditioner bars.
- AminoSensyl™ HC and Emulsense™ HC: Naturally derived, conditioning alternatives for solid formats.
Table: Common Surfactants Used in Skincare and Haircare Formulations
Surfactants come in many forms, some derived from plants, others synthetically produced. Each has unique cleansing, foaming, and conditioning properties that make it suitable for specific skincare and haircare formulations. The table below highlights a variety of commonly used surfactants, organized by their charge type, origin, and typical functions.
Whether you’re developing a sulfate-free shampoo, a gentle facial cleanser, or a conditioning bar, understanding these ingredients helps you create products that clean effectively while respecting the skin and scalp barrier.
| Charge Type | Surfactant Name | Source | Typical Use & Notes |
| Anionic | Disodium Laureth Sulfosuccinate | Synthetic | Mild cleansing agent, less irritating than SLS; used in shampoos and body washes. |
| Anionic | Potassium Cocoate | Naturally Derived | Derived from coconut oil; gentle cleanser often used in liquid soaps. |
| Anionic | Sodium C14-16 Alpha Olefin Sulfonate | Naturally Derived | Strong degreasing power; used in clarifying shampoos. |
| Anionic | Sodium Cocoyl Glutamate | Naturally Derived | Mild surfactant with good foam; biodegradable. |
| Anionic | Sodium Cocoyl Isethionate (SCI) | Naturally Derived | Creamy lather; sulfate-free; ideal for solid shampoos and syndet bars. |
| Anionic | Sodium Coco-Sulfate (SCS) | Naturally Derived | High foaming, effective cleanser, slightly gentler than SLS. |
| Anionic | Sodium Lauroyl Glutamate | Naturally Derived | Mild, amino acid-based cleanser; suitable for sensitive skin. |
| Anionic | Sodium Lauryl Sulfate (SLS) | Synthetic | Strong cleanser and foamer; may cause dryness in some users. |
| Anionic | Sodium Laureth Sulfate (SLES) | Synthetic | Most common high-foam anionic; milder than SLS; shampoos and washes. |
| Anionic | Sodium Lauroyl Methyl Isethionate (SLMI) | Naturally Derived | Liquid-friendly SCI alternative; sulfate-free, mild cleansers. |
| Anionic | Sodium Cocoyl Glycinate | Naturally Derived | Very mild, luxurious foam. |
| Anionic | Sodium Lauroyl Sarcosinate | Naturally Derived | Mild amino-acid surfactant; sulfate-free; sensitive skin cleansers. |
| Anionic | Sodium Lauryl Sulfoacetate (SLSa) | Naturally Derived | Milder alternative to SLS; good lather and mild cleansing. |
| Amphoteric | Coco-betaine | Naturally Derived | Balances cleansing and mildness; foam booster. |
| Amphoteric | Cocamidopropyl Betaine | Naturally Derived | Very common secondary surfactant; reduces irritation of anionics. |
| Amphoteric | Lauryl Betaine | Naturally Derived | Similar to coco-betaine; often used in shampoos and facial cleansers. |
| Amphoteric | Disodium Cocoamphodiacetate | Naturally Derived | Ultra-mild co-surfactant; baby shampoos, micellar products |
| Cationic | Behentrimonium Chloride | Synthetic | Conditioning agent; reduces static; used in conditioners. |
| Cationic | Behentrimonium Methosulfate (BTMS) | Naturally Derived | Excellent detangler; adds slip in solid conditioner bars. |
| Cationic | Brassicyl Isoleucinate Esylate | Naturally Derived | Plant-based conditioning surfactant; mild and biodegradable. |
| Cationic | Cetrimonium Chloride | Synthetic | Antistatic and conditioning; common in rinse-off conditioners. |
| Cationic | Distearoylethyl Dimonium Chloride | Naturally Derived | Used for conditioning and softening; in solid haircare bars. |
| Nonionic | Caprylyl/Capryl Glucoside | Naturally Derived | Mild cleanser and solubilizer; used in micellar waters. |
| Nonionic | Coco Glucoside | Naturally Derived | Gentle, biodegradable; stabilizes foam in mild cleansers. |
| Nonionic | Decyl Glucoside | Naturally Derived | Very mild; ideal for baby products and sensitive skin. |
| Nonionic | Lauryl Glucoside | Naturally Derived | Provides creamy texture; supports foam stability. |
| Nonionic | Methyl Glucamide | Naturally Derived | Conditioning properties; often used in mild shampoos. |
| Nonionic | Sucrose Cocoate | Naturally Derived | Very mild surfactant and emollient; used in skin cleansers. |
Note:
Some surfactants listed here as “naturally derived” also have synthetic versions available in the market. If you plan to make claims such as natural, naturally derived, or organic, always verify your supplier documentation and certifications to ensure compliance with relevant standards and labeling requirements.
The Truth About SLS: Is It Really That Bad?
Sodium Lauryl Sulfate, better known as SLS, has become one of the most controversial surfactants in cosmetic formulations. You’ll find it in everything from shampoos and cleansers to toothpaste. It’s also frequently targeted by “free from” marketing campaigns, often portrayed as a harsh or even toxic ingredient. But does SLS really deserve its bad reputation?
What Science Says About SLS Safety
SLS is a powerful anionic surfactant, valued for its strong cleansing and high-foam performance. It effectively lifts dirt, excess oil, and buildup. Regulatory agencies including the EU Scientific Committee on Consumer Safety and the U.S. FDA have concluded that SLS is safe for rinse-off products when used within recommended concentrations.
However, prolonged contact or high concentrations can disrupt the skin barrier and cause irritation, especially for people with sensitive skin or compromised scalp health. This is why you’ll often see lower percentages in facial cleansers compared to shampoos or body washes.
Common Myths About SLS-Free Labels
Many brands proudly display “SLS-Free” on their packaging. While this can be helpful for consumers who prefer milder cleansers, it’s important to understand what SLS-free actually means:
It doesn’t automatically mean the product is gentler, some alternatives can be just as cleansing.
SLS-free formulas often still use other sulfates, like Sodium Coco-Sulfate or SLES (Sodium Laureth Sulfate).
Sulfate-free doesn’t always mean soap-free, or that the product has no synthetic ingredients.
Choosing sulfate-free is mainly about reducing potential irritation and meeting consumer expectations, not about avoiding something inherently toxic.
When to Choose SLS-Free Alternatives
- You might prefer SLS-free surfactants if:
- You have sensitive skin or a dry, flaky scalp.
- You’re formulating products intended for daily use.
- Your brand positioning emphasizes mild, eco-friendly, or natural-derived solutions.
- You’re targeting consumers concerned about “clean beauty.”
Milder alternatives—such as Sodium Cocoyl Isethionate (SCI) or Sodium Lauroyl Sarcosinate can deliver effective cleansing without the same degree of potential irritation.
How to Choose the Right Surfactant for Your Formulations
Choosing a surfactant isn’t just about big foam or a natural-sounding name. A smart selection balances cleansing performance, skin and scalp tolerance, texture, and the story you want to tell about your product. Here’s what to consider as you develop your formula.
Factors to Consider: Skin Type, Hair Type, and Product Use
Who is it for?
- Sensitive or easily irritated skin?
Go for nonionic surfactants (like glucosides) or mild amphoteric surfactants (like cocamidopropyl betaine). These are less likely to strip natural oils or disrupt the skin barrier.
- Oily scalp or heavy buildup?
Use anionic surfactants with stronger cleansing, such as Sodium Lauryl Sulfoacetate (SLSa) or Sodium Coco-Sulfate (SCS). These help remove excess sebum and styling residue.
- Dry, curly, or color-treated hair?
Choose sulfate-free anionics like SCI or glutamates, combined with conditioning agents to prevent dryness and help detangling.
How often will it be used?
- Daily products should stay on the gentle side, with mild surfactants and lower active concentrations.
- Weekly clarifying shampoos can use stronger cleansers since they aren’t applied frequently.
What’s the product format?
- Solid bars need surfactants in powder or noodle form that bind well. SCI is a top choice here for mildness and creamy lather.
- Liquid cleansers often rely on Decyl Glucoside or Coco Glucoside, which are gentle and easy to formulate but may need thickeners.
Tips for Creating Mild, Skin-Friendly Cleansers
- Blend surfactants: Combining anionic and amphoteric types reduces irritation without sacrificing cleaning power. For example, pairing SCS with cocamidopropyl betaine softens the formula’s impact on skin.
- Check the pH: Aim for pH 4.5–5.5 to protect the acid mantle and avoid dryness.
- Use the right concentration: High levels, even of mild surfactants, can still irritate. Always formulate with the lowest effective dose.
- Add soothing ingredients: Panthenol, allantoin, or botanical extracts help calm and moisturize skin after cleansing.
Sustainable and Biodegradable Choices
If you want your products to align with green beauty principles, focus on surfactants that are:
- Sourced from renewable materials like coconut, corn, or sugar.
- Easily biodegradable and low-impact on waterways.
- Approved by standards such as COSMOS or ECOCERT.
Good examples include:
- Decyl Glucoside: Plant-based, very mild, highly biodegradable.
- Sodium Cocoyl Glutamate: Amino acid–based, gentle, and environmentally friendly.
- Coco Glucoside: Balances mild cleansing with rich foam and a solid sustainability profile.
In short, the best surfactant is the one that fits your target user, your product claims, and your performance goals, without compromising skin health or the environment.
Final Thoughts
Surfactants are some of the most essential, and misunderstood, ingredients in skincare and haircare formulations. From the powerful cleansing of anionics to the soft conditioning of cationics, there’s no one-size-fits-all solution. The key is balance: choosing surfactants that meet your performance needs without compromising the health of the skin, hair, or scalp.
If you’re formulating for sensitive users, or simply want to create a gentler product line, it’s worth investing the time to explore mild, sulfate-free, and biodegradable options. And remember: the most successful products don’t just clean, they create a pleasant experience that keeps customers coming back.
Explore More: Download Your Haircare Formulation Guide
Want to dive deeper into surfactants, conditioning agents, and how to develop professional-grade shampoo and conditioner bars?
🎁 Download our Solid Shampoo & Conditioner Formulation Guide, packed with detailed ingredient breakdowns, formulation tips, and step-by-step recipes to help you create high-performance solid haircare products from scratch.
Whether you’re a passionate DIY formulator or launching your own brand, this free resource will inspire you to craft products that are as effective as they are gentle.
